OptiLIF® Procedure
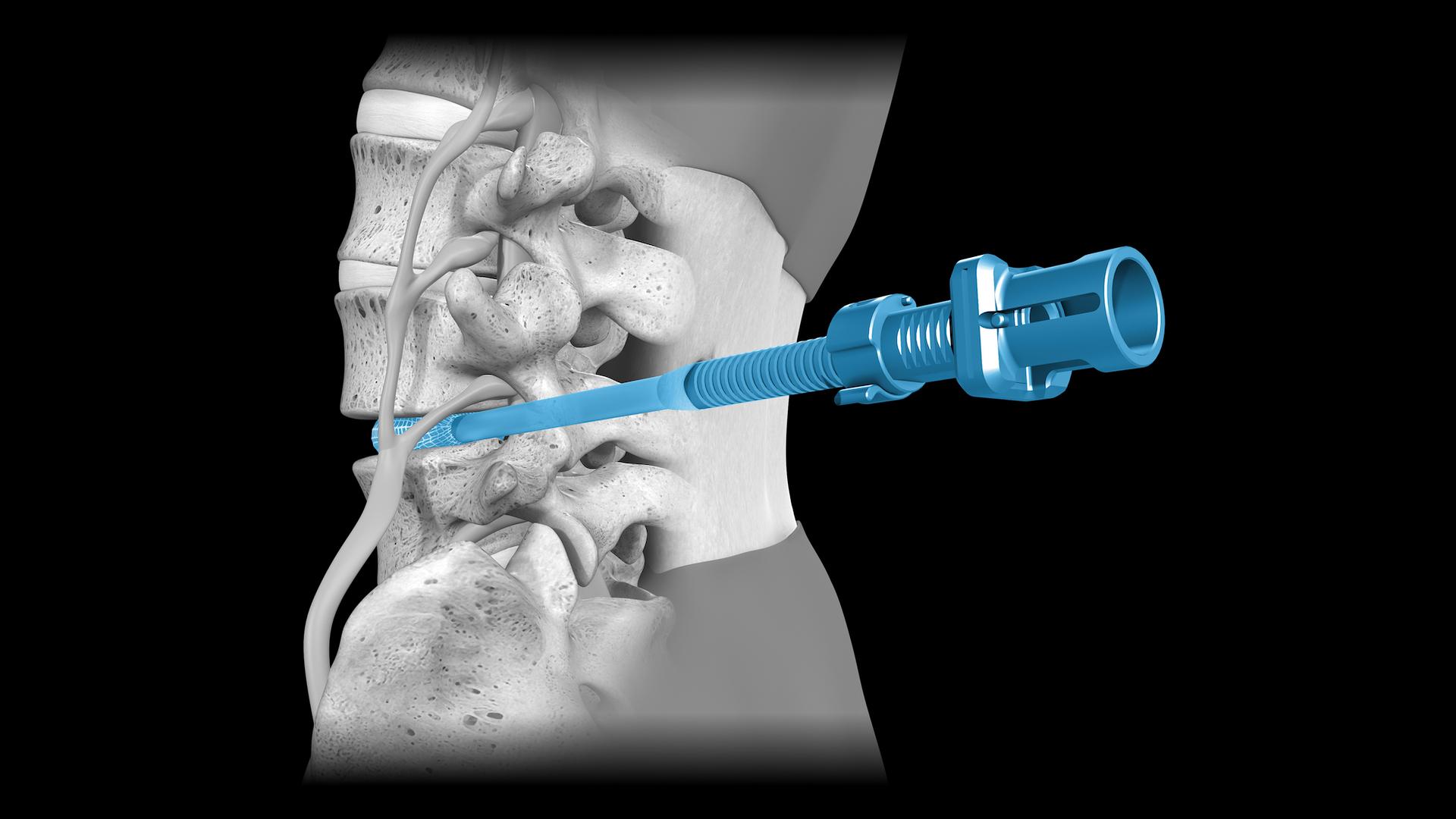
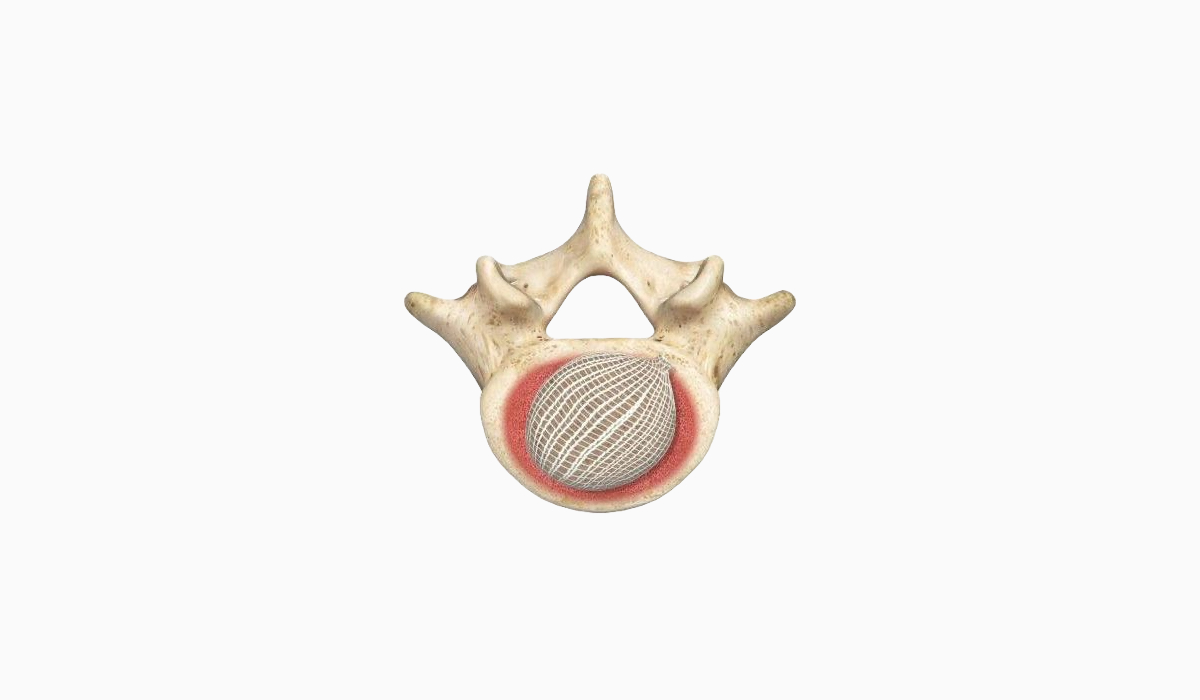
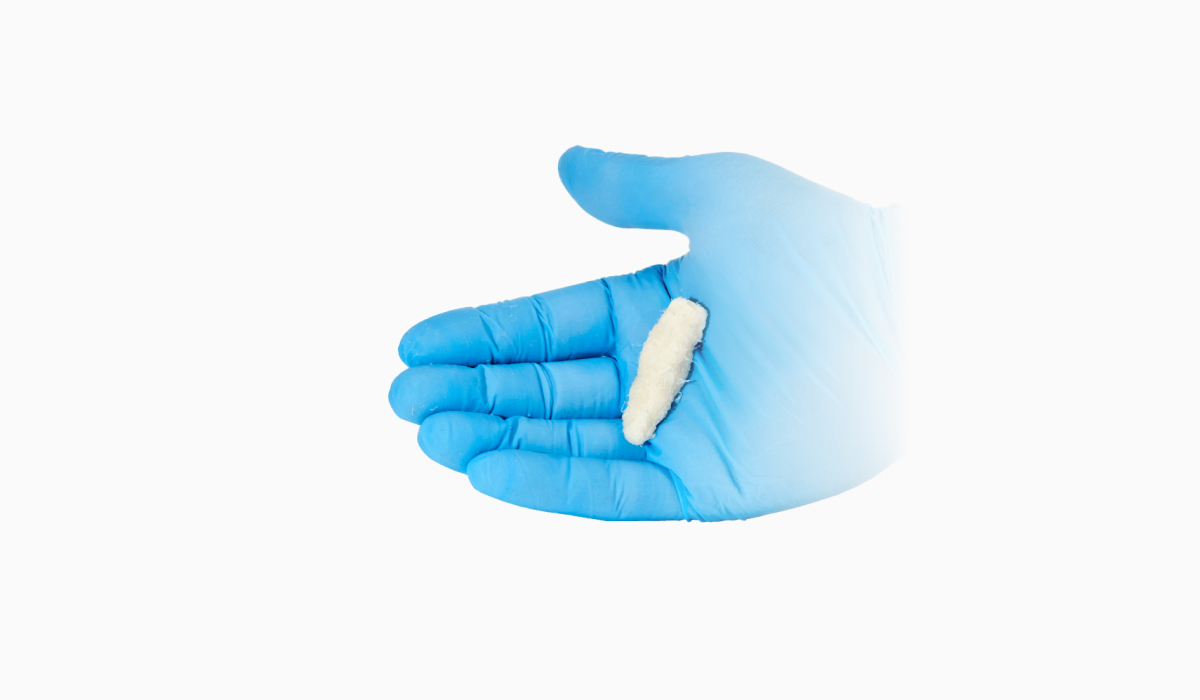
The OptiLIF procedure is one of the least invasive lumbar interbody fusion procedures currently available and features Spineology’s proprietary OptiMesh® implant. The procedure is completed through a 6.6 mm ID tubular retractor and uses proprietary instrumentation to perform a robust discectomy, prepare the endplates, and place OptiMesh, the only conforming, patient specific expandable implant that results in ALIF-size implant.